Graphite, Properties, Uses, Structure
4.7 (717) · € 21.00 · En Stock
Graphite, mineral consisting of carbon. Graphite has a layered structure that consists of rings of six carbon atoms arranged in widely spaced horizontal sheets. Graphite thus crystallizes in the hexagonal system, in contrast to the same element crystallizing in the octahedral or tetrahedral system
.jpg)
Differences Between Graphene and Graphite

Allotropes of carbon - Wikipedia

DIAMOND Vs GRAPHITE Diamond and - Kannan's Chemistry page

What is Graphite: Definition, Structure, Properties, Process, Uses

Graphene and its Applications WORLD CONGRESS ON MATERIALS SCIENCE AND ENGINEERING

Graphite - Wikipedia
Open Knowledge Wiki - What is Graphite
Graphite - Structures - (CCEA) - GCSE Combined Science Revision - CCEA Double Award - BBC Bitesize

STRUCTURE, PROPERTIES & USES OF GRAPHITE

Graphene synthesis, characterization and its applications: A review - ScienceDirect

Graphite is carbon? Carbon graphite properties, use, structure, and diamond - DanCrabon
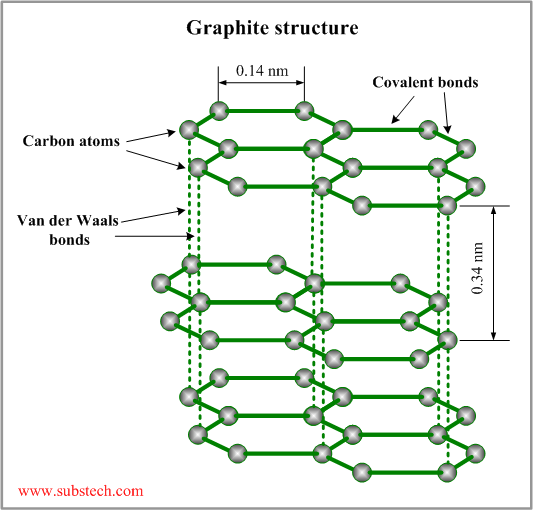
Graphite [SubsTech]
.png)
Single-walled Carbon Nanotubes: Structure, Properties, Applications












